What Is The Gas Makeup Of Air
Three-fourths of all air resides in the troposphere, the everyman layer of the Earth's atmosphere. Air is a mixture of gases, almost of which are naturally occurring. Air also contains a significant amount of human-made air pollutants, including some that are not safe to breathe and some that warm our planet's climate. The troposphere likewise contains water in all three phases (liquid, solid, and gas) likewise equally solid particles, chosen aerosols.

The dry composition of the atmosphere is mostly nitrogen and oxygen. Information technology too contains partial amounts of argon and carbon dioxide and trace amounts of other gases, such as helium, neon, methane, krypton, and hydrogen (NASA).
UCAR
Gases
The well-nigh arable naturally occurring gas is Nitrogen (N2), which makes up about 78% of air. Oxygen (O2) is the second almost arable gas at about 21%. The inert gas Argon (Ar) is the third most abundant gas at .93%. At that place are also trace amounts of carbon dioxide (CO2), neon (Ne), helium (He), methyl hydride (CH4), krypton (Kr), hydrogen (H2), nitrous oxide (NO), xenon (Xe), ozone (O3), iodine (I2), carbon monoxide (CO), and ammonia (NH3) in the temper.
H2o Vapor
Due to the water cycle, the amount of water in the air is constantly changing. The lower troposphere can contain up to 4% water vapor (H2o) in areas near the torrid zone, while the poles contain but trace amounts of water vapor. The concentration of h2o vapor decreases drastically with altitude. The upper troposphere has considerably less water vapor than air near the surface, the stratosphere and mesosphere take near no water vapor, and the thermosphere contains none at all.
Aerosols
Air also contains tiny solid particles called aerosols, such as dust, bounding main table salt, and ash from erupting volcanoes or forest fires. Many of these particles are so pocket-size that they are microscopic. Others are big plenty to run across. Aerosols affect climate by helping clouds form and shading the planet by scattering or arresting sunlight. In the last century, manufacturing and widespread utilise of combustion engines take increased the number of aerosols in the temper as particulate matter spews from smokestacks and exhaust pipes. Burning wood and other materials also add particles to the air.
Atmospheric Chemistry
Like everything on Earth, the air is made of chemicals. The chemicals in the air often combine with each other, or with other chemicals from the Earth'southward surface, through chemical reactions. Many of these chemic reactions assist maintain salubrious natural environments and are vital for plants and animals. Nitrogen gas in the atmosphere does nearly null, but nitrogen elsewhere on Earth is essential for life. Through the nitrogen cycle, nitrogen makes its fashion into soil and water, binds with other elements, and can be used by living things. Oxygen from the atmosphere causes oxidation reactions that help break down matter and release nutrients into soils, and is used past humans and animals in cellular respiration.
Atmospheric chemistry in the troposphere is also influenced by human being-fabricated chemicals that tin can negatively bear on human health and the environment. For case:
- Vehicle exhaust contains nitrogen dioxide, as well as other polluting chemicals such as carbon monoxide and sulfur dioxide. Nitrogen dioxide reacts with atmospheric oxygen to form tropospheric ozone which is chancy to plant and animal cells.
- Smog, which is mainly made of ozone and particulate carbon (soot) emitted past coal-burning ability plants, causes damage to the lungs of humans and animals.
- Factories that burn fossil fuels also release sulfur and nitrogen dioxides, which combine with water in the atmosphere to make acid rain. Acid rain causes damage to natural and human being-fabricated environments.
Chemistry of the Air
The tabular array below lists the major gas components and their part in the atmosphere. Click on each molecule name to acquire more about them.
| Gas | Chemical & Molecular Construction | Role in the Atmosphere |
|---|---|---|
| Nitrogen | 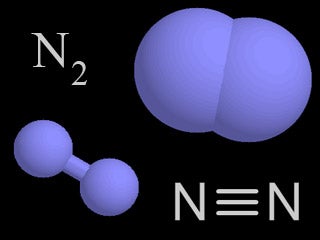 | 78% of the air in the atmosphere is nitrogen. Nitrogen is transferred to plants, animals, and the environment through the nitrogen bicycle. |
| Nitrogen Oxides | 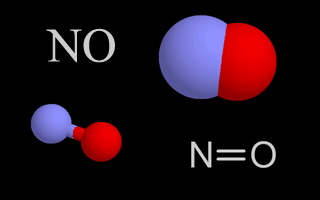 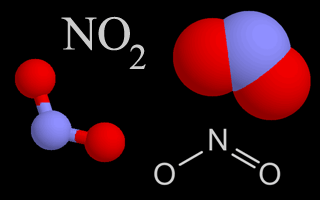 | Nitrogen oxides are air pollutants that contribute to the formation of ozone. They also create nitric acid, which is part of acid rain, when they mix with water droplets in the air. |
| Oxygen | 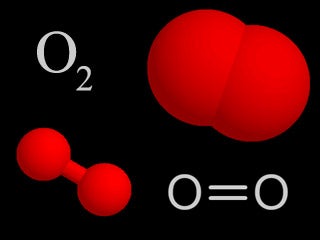 | Oxygen makes up 21% of the temper. Information technology is highly reactive and forms compounds with numerous other chemicals, and is necessary for respiration in living things. |
| Ozone | 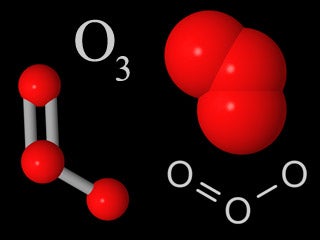 | Ozone in the troposphere is a human-made pollutant. Ozone in the stratosphere forms the ozone layer, which is crucial for the survival of life at the Earth'south surface. |
| Argon |  | Argon makes upwardly near 1% of the atmosphere and comes by and large from the decay of potassium in the Earth's crust. It is an inert gas, which means that information technology does non react with other chemicals. |
| Water Vapor | 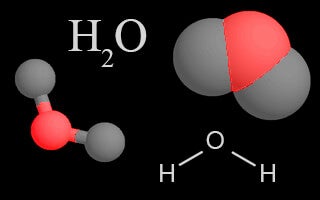 | Water is cycled through all of Earth'due south systems in each of its three phases: solid, liquid, or gas. Water vapor in the atmosphere is a greenhouse gas due to its oestrus-trapping ability. |
| Carbon Dioxide | 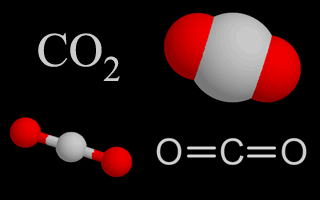 | Carbon dioxide naturally composes almost .03% of the atmosphere, but the amount is increasing due to the burning of fossil fuels. Plants and eubacteria use carbon dioxide during photosynthesis. Humans, other animals, and plants add it to the air through respiration. Carbon dioxide is a heat-trapping greenhouse gas. |
| Carbon Monoxide | 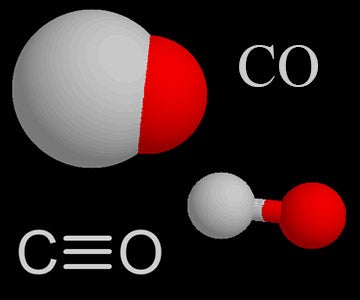 | Carbon monoxide in the air comes from burning fuel in vehicles, volcanoes, and forest fires. It is a poisonous gas. |
| Methane | 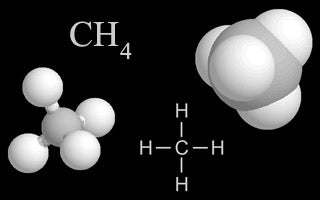 | Methane gas is released into the air from landfills, livestock and their manure, and from oil and gas wells. It is also created when organic textile decomposes. It is a heat-trapping greenhouse gas. |
| Sulfur Oxides | 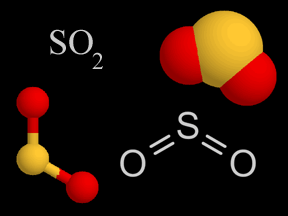 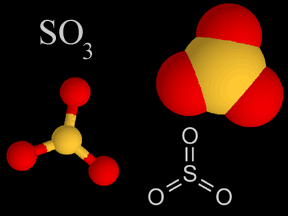 | Sulfur oxides are produced when coal and oil are burned. It'south also released from volcanoes. The sulfur oxides mix with water droplets in the atmosphere to create sulfuric acid, which is a component of acrid rain. |
© 2020 UCAR
Source: https://scied.ucar.edu/learning-zone/air-quality/whats-in-the-air
Posted by: dangeloancell1948.blogspot.com

0 Response to "What Is The Gas Makeup Of Air"
Post a Comment